 In galvanic cells, copper is a cathode which is dipped in CuSO4 solution.Ĭold cathodes: Cathodes that are not electrically heated by a filament. This means the cathode gains electrons from the external circuit and gets reduced. Generally, the cathode is the electrode where reduction reaction takes place. This is common in an electrochemical cell. It is a point where reduction takes place. The current through a battery flows in an opposite direction of the current during discharge in the recharging battery.Ī cathode is a place from where electricity leaves through a cell. The anode is the positive terminal that receives current from an external generator in a recharging battery. A positively charged platinum electrode where H2 gas is oxidized to H+ ions is the anode. Additionally, an anode can be a plate or wire having an excess positive charge.Ī simple example is electrolysis of water. In an electrolytic cell, it is again positive. If we take a galvanic cell, the anode is negative in nature and the electrons mostly move towards the external part of the circuit. These electrons then move up and into the driving circuit. Generally, at an anode, negative ions or anions due to its electrical potential tend to react and give off electrons. In the most basic form, an anode in electrochemistry is the point where an oxidation reaction occurs. Anodes react and give off the electrons by which they can go into the driving circuit. It is a point where oxidation takes place. The anode cathode symbol respectively are A and K.ĭifference Between Cyclic and Noncyclic Photophosphorylationĭifference Between Cytoplasm and ProtoplasmĪn anode is a place from where electricity passes through in a cell. The anode and cathode charge are positive and negative respectively. Likewise, an anode is an electrode from which a current enters into a polarized electrical device.Īnode and cathode are the two types of electrodesĪn anode is an electrode from which polarized current enters the outer circuit.Ī cathode is an electrode from which a conventional current leaves a polarized electrical device. Therefore, a cathode is an electrode from which the current exits a polarized electrical device. 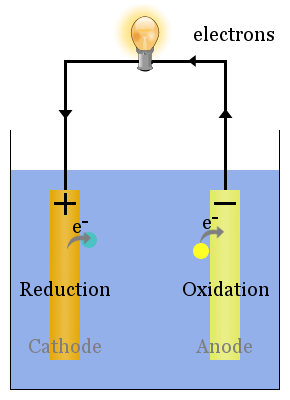 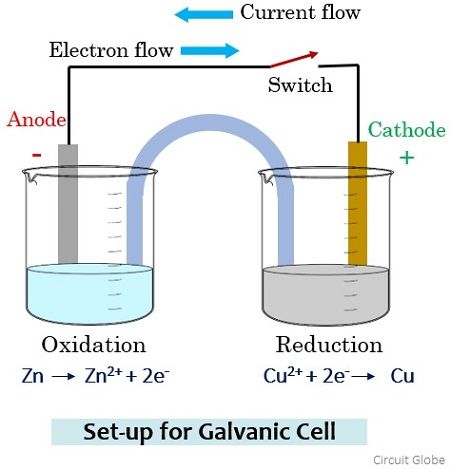
Cathode and Anode are both defined by the flow of current. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
